Navigating Medical Device Registration in India: A Comprehensive Guide
- Admirus

- Aug 14, 2023
- 3 min read

Healthcare industry in India is rapidly evolving, with innovations in medical devices playing a pivotal role in improving patient care and outcomes.
However, before these cutting-edge devices can be used, they must go through a thorough regulatory process known as Medical Device Registration.
Understanding the Significance
Medical device registration is a crucial step to ensure the safety, quality, and efficacy of medical devices in the Indian market. The regulatory process ensures that only devices meeting the necessary standards and requirements are made available to healthcare professionals and patients. This process not only safeguards the health and well-being of the population but also fosters confidence in the industry.
Key Steps in Medical Device Registration
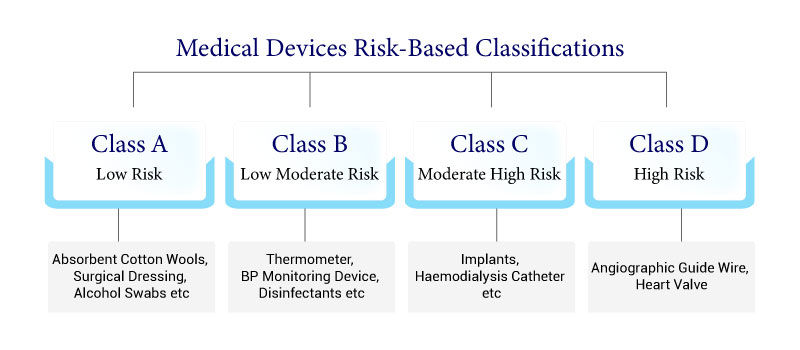
Classification: The first step is to determine the appropriate classification of the medical device as per the Indian regulatory framework. Devices are categorized based on their risk level and intended use.
Preparation of Documentation: Detailed documentation, including technical specifications, clinical data (if required), labeling, and manufacturing information, must be prepared meticulously. This documentation is the foundation for the registration process.
CDSCO Submission: The Central Drugs Standard Control Organization (CDSCO) is the primary regulatory authority responsible for medical device registration in India. The submission of the required documents to CDSCO is a crucial step in the process.
Review and Approval: CDSCO reviews the submitted documents to ensure they comply with Indian regulations. This may involve thorough scrutiny of clinical data, safety information, and manufacturing practices.
Testing and Inspections: Depending on the device's classification, laboratory testing and inspections may be required to verify its safety and efficacy.
Grant of Registration: Upon successful completion of the review process, CDSCO grants the registration, allowing the device to be legally marketed and distributed in India.
The Central Drugs Standard Control Organisation (CDSCO)

The Central Drugs Standard Control Organisation (CDSCO) is the esteemed National Regulatory Authority (NRA) of India, operating under the Directorate General of Health Services within the Ministry of Health & Family Welfare, Government of India. Headquartered at FDA Bhawan, Kotla Road, New Delhi, CDSCO is a pivotal institution with an extensive national presence.
With six zonal offices, four sub-zonal offices, thirteen port offices, and seven laboratories spanning the country, it effectively oversees the safety, efficacy, and quality of medical products.
Founded on the cornerstone of the Drugs & Cosmetics Act, 1940, and its corresponding rules from 1945, CDSCO bears a crucial responsibility for the regulation of drugs and cosmetics in India. Its primary goal is to ensure the well-being of patients by uniformly implementing the provisions of the Act and its rules. Transparency, accountability, and uniformity are the guiding principles behind CDSCO's mission, a testament to its dedication to the safety and efficacy of medical products manufactured, imported, and distributed within the country.
CDSCO operates as the pivotal entity responsible for various critical tasks. It approves drugs, oversees the conduct of clinical trials, sets high standards for drugs, monitors the quality of imported drugs, and coordinates the activities of State Drug Control Organizations. Its expertise plays a crucial role in fostering consistency in enforcing the Drugs and Cosmetics Act, thereby safeguarding public health.
In partnership with state regulators, CDSCO jointly grants licenses for specialized categories of critical drugs, which includes blood and blood products, I.V. fluids, vaccines, and sera. Through these collective efforts, CDSCO contributes significantly to the medical safety landscape, ensuring that essential medical products meet the highest standards and are available to benefit the people of India.
Role of Regulatory Bodies
Apart from CDSCO, there are other bodies, such as the National Medical Devices Promotion Council (NMDPC), that play a role in shaping the regulatory landscape for medical devices in India. These organizations work to streamline the registration process, promote innovation, and ensure adherence to the highest quality standards.
Medical device registration in India is a comprehensive process designed to ensure the safety and efficacy of medical devices available in the market. It is crucial for manufacturers and importers to understand the regulatory requirements and comply with them. By navigating this process diligently, we contribute to advancing healthcare, fostering innovation, and improving patient outcomes in India.
In the dynamic landscape of medical device registration in India, where adherence to regulatory standards is paramount, Admirus emerges as the trusted partner for entrepreneurs and companies. With our deep understanding of the CDSCO's requirements, coupled with our expertise in navigating the intricate regulatory process, we stand ready to assist you in obtaining timely and successful registrations. Admirus simplifies the complex journey, ensuring compliance, transparency, and swift approvals, so you can focus on innovation and bring your medical devices to market with confidence.

.png)
Comments